Chemistry Journal of Moldova
Physical chemistry and chemical physics
Author(s):
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2014 Volume 9, no.1
Pages: 128-131
Ion Geru, Olga Bordian, Constantin Loshmansky, Ion Culeac, Constantin Turta
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2014 Volume 9, no.1
Pages: 128-131
Full Text (PDF): Download
DOI: dx.doi.org/10.19261/cjm.2014.09(1).17
Graphical Abstract: In this paper we present experimental results on preparation and characterization of colloidal CdSe quantum dots in organic solvent. CdSe QDs were synthesized following a modified literature method.
CdSe QDs were isolated by adding acetone to the cooled solution followed by centrifugation.
CdSe QDs have been characterized by UV-Vis absorption and photoluminescent (PL) spectroscopy. The average CdSe particles size estimated from the UV-Vis absorption spectra was found to be in the range 2.28-2.92 nm which is in good agreement with PL measurements.
Downloads: 98
Author(s):
Field: Physical chemistry and chemical physics
Type: Review
Issue: 2013 Volume 8, no.2
Pages: 32-41
Lilia Anghel, Gheorghe Duca
Field: Physical chemistry and chemical physics
Type: Review
Issue: 2013 Volume 8, no.2
Pages: 32-41
Full Text (PDF): Download
DOI: dx.doi.org/10.19261/cjm.2013.08(2).03
Graphical Abstract: Iron-based nanoparticles have gained a lot of attention due to their properties which offer a broad range of biomedical and industrial applications. Traditional methods of synthesis of iron nanoparticles strongly influence their properties and limit their applicability. Recently, there has been a growing interest in the development of biological routes of syntheses of iron nanoparticles as the resulting particles have structural characteristics required by biomedical field. The mechanism for the synthesis of iron-based nanoparticles by microorganisms and its current limitation are presented.
Downloads: 50
Author(s):
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2013 Volume 8, no.1
Pages: 90-94
N. Timbaliuc, T. Lupascu
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2013 Volume 8, no.1
Pages: 90-94
Full Text (PDF): Download
DOI: dx.doi.org/10.19261/cjm.2013.08(1).11
Graphical Abstract: The adsorption properties of activated carbons, obtained from local raw materials (nut shells, peach and plum stones), towards Congo Red and vitamin B12 have been studied. The values of adsorption of these marker-substances are in direct correlation with the structural characteristics of the studied samples of activated carbons, in particular, with their mesopore volume.
Downloads: 31
Author(s):
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2013 Volume 8, no.1
Pages: 95-100
O.G. Kolumbin, L.N. Ognichenko, A.G. Artemenko, P.G. Polischuk, М.A. Kulinsky, Е.N. Мuratov, V.E. Kuz’min, V.A. Bobeica
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2013 Volume 8, no.1
Pages: 95-100
Full Text (PDF): Download
DOI: dx.doi.org/10.19261/cjm.2013.08(1).12
Graphical Abstract: In our study the dataset containing 489 pesticides and their active substances of different classes of organic compounds was used for analysis. For compounds of analyzed dataset the values of lipophilicity, water solubility, toxicity, bioavailability and mutagenicity were predicted by developed QSAR models. The most environmentally hazardous substances were identified using prediction of QSAR models for pesticides. The satisfactory coincidence between the experimental values of investigated properties and their predicted values by QSAR models was obtained (coefficient of determination in the range 83-94 %).
Downloads: 36
Author(s):
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2012 Volume 7, no.2
Pages: 143-146
E. Pylypchuk, V. Mishchenko and T. Yu. Gromovoy
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2012 Volume 7, no.2
Pages: 143-146
Full Text (PDF): Download
DOI: dx.doi.org/10.19261/cjm.2012.07(2).02
Graphical Abstract: It was shown by X-ray phase analysis, IR spectra analysis and MALDI-ToF mass spectrometry methods that interaction of synthetic hydroxyapatite with a solution of immunoglobulin G leads to its partial dissolution due to leaching from the surface of calcium triphosphate which, in our opinion, forms complexes with immunoglobulin G.
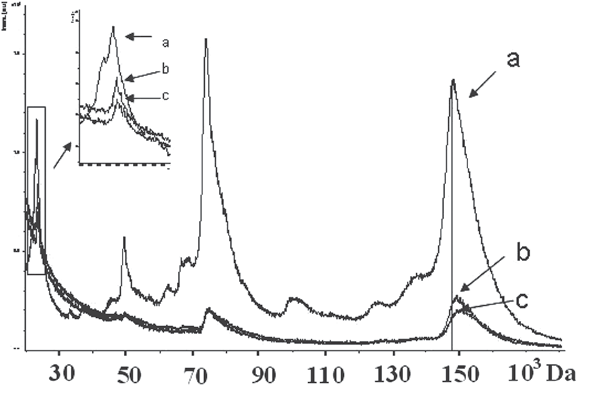
Downloads: 31
Author(s):
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2010 Volume 5, no.2
Pages: 68-72
Ion Dranca, Nicon Ungur,Tudor Lupascu, Oleg Petuhov
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2010 Volume 5, no.2
Pages: 68-72
Full Text (PDF): Download
DOI: dx.doi.org/10.19261/cjm.2010.05(2).09
Graphical Abstract: The methods of thermogravimetry (TGA) and differential scanning calorimetry (DSC) have been used to study the thermal and thermo-oxidative degradation of polystyrene (PS) and a PS-clay nanocomposite. An advanced isoconversional method has been applied for kinertic analysis. Introduction of the clay phase increasers the activation energy and affects the total heat of degradation, which suggests a change in the reaction mechanism. The obtained kinetic data permit a comparative assessment of the fire resistance of the studied materials.
Downloads: 25
Author(s):
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2010 Volume 5, no.2
Pages: 73-82
Vasilii Gutsanu, Constantin Turta, George Filoti
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2010 Volume 5, no.2
Pages: 73-82
Full Text (PDF): Download
DOI: dx.doi.org/10.19261/cjm.2010.05(2).10
Graphical Abstract: Using the Mossbauer spectroscopy and other physical methods it was demonstrated the presence of different
Downloads: 16
Author(s):
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2010 Volume 5, no.2
Pages: 83-87
Secara Natalia
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2010 Volume 5, no.2
Pages: 83-87
Full Text (PDF): Download
DOI: dx.doi.org/10.19261/cjm.2010.05(2).11
Graphical Abstract: The reaction of dihydroxyfumaric acid with the free radical 2,2-diphenyl-1-picrylhydrazyl (DPPH) was studied using the stopped-flow method, in order to describe the reaction kinetics. Dihydroxyfumaric acid reacts very rapidly with DPPH, the reaction being completed in several minutes. This 2-stoichiometric reaction proceeds in two stages, with reaction orders of 1 and 0.76 with respect to DPPH, and 0.5 and 0.3 with respect to DHF, respectively. The rate constant of the two stages of the reaction were found to be 39.1 (L/mol•s) and 0.0012 (s-1) at 20º C and pH 4.0.
Downloads: 36
Author(s):
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2011 Volume 6, no.2
Pages: 84-90
Iolanta I. Balan, Natalia N. Gorinchoy
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2011 Volume 6, no.2
Pages: 84-90
Full Text (PDF): Download
DOI: dx.doi.org/10.19261/cjm.2011.06(2).18
Graphical Abstract: The four-stage mechanism of reaction of the rhodium trihydride complex [(triphos)RhH3] (triphos=1,1,1-tris(diphenylphosphanylmethyl)ethane) with the white phosphorus molecule resulting in the phosphane and the cyclo-P3 complex [(triphos)M(η3-P3] is analyzed on the basis of ab initio calculations of reactants, products, and intermediate complexes of reaction. It is shown that generation of the transient complex [(triphos)RhH(η1:η1-P4)] followed by intramolecular hydrogen atom migration from the metal to one of the phosphorus atoms is the energetically favourable process. Calculations also show that P4 molecule is activated by coordination to the above complex: the metal-bonded P-P edge is broken, and the tetrahedron P4 is opened to form the butterfly geometry. This activation is realized mainly due to the one-orbital back donation of 4d-electron density from the atom of Rh to the unoccupied antibonding triple degenerate t1*-MO of P4.
Downloads: 26
Author(s):
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2011 Volume 6, no.2
Pages: 91-95
Ion Dranca, Igor Povar and Tudor Lupascu
Field: Physical chemistry and chemical physics
Type: Research paper
Issue: 2011 Volume 6, no.2
Pages: 91-95
Full Text (PDF): Download
DOI: dx.doi.org/10.19261/cjm.2011.06(2).19
Graphical Abstract: This research has been carried out in order to demonstrate the use of differential scanning calorimetry (DSC) in detecting and measuring α- and β-relaxation processes in amorphous pharmaceutical systems. DSC has been employed to study amorphous samples of poly (vinylpyrrolidone) (PVP), indomethacin (InM), and ursodeoxycholic acid (UDA) that are annealed at temperature (Ta) around 0.8 of their glass transition temperature (Tg). Dynamic mechanical analysis (DMA) is used to measure β- relaxation in PVP. Yet, the DSC has been used to study the glassy indomethacin aged at 0 and -10 oC for periods of time up to 109 and 210 days respectively. The results demonstrate the emergence of a small melting peak of the α-polymorph after aging for 69 days at 0°C and for 147 days at -10°C (i.e., ~55°C below the glass transition temperature) that provides evidence of nucleation occurring in the temperature region of the β-relaxation.
Downloads: 23






